22+ 6.02 x10 23 calculator
6020000000000000000000006 Multiply 602 and 100000000000000000000000 to get. If you were to write this in a calculator it would be 602E23.

Sections 10 1 10 2 Mole Conversions 1 Mole 6 02x10 23 Atoms Molar Mass G 22 4 L Molecules Of Any Gas Formula Units At Stp Mass Mole Problem Ppt Download
1 mole of any substance has 602 x 1023 molecules.

. The mass in grams of 602 x 10 23 particles of a substance is now called the molar mass mass of 100 mole. Type in the significant figures 602. Hit the equals button.
In either case what this is really saying is that you move the. The energy required to break one mole of Cl - Cl bond is 242 kJmol. So in our case we have 1 mole of water.
In this calculator numbers in scientific notation must be entered in e notation. Now a one mole quantity specifies 6022 1023 formula. For example 1234 x.
Calculate 10 to the power of 23 and get 100000000000000000000000. Answer 1 of 4. So 2645 x 602 x 1023 will be roughly 15 x.
What is the mass of 602 x10. Hit the 2nd key top left 3. In this calculator numbers in scientific notation must be entered in e notation.
6022 1023 6022 10 23. Which mass is made up of 602 x10 23 atoms. Since the exponent of the scientific notation is positive move the decimal point 23 23 places to the right.
This is another way of writing scientific notation. How to use this calculator. Solve advanced problems in Physics Mathematics and Engineering.
For example 1234 x. The 1 mole of substance is equal to 6022140857 x 1023 units of substance such as atoms. Free Online Scientific Notation Calculator.
How to use this calculator. If you divide the charge on a mole of electrons by the charge on a single electron you obtain a value of Avogadros number of 602 x 1023 particles per mole. The concept of moles to atoms conversion is totally dependent upon Avogadros number.
Convert to Regular Notation 60221023. I guess that the problem might be in entering the exponent but a bit of mental arithmetic shows that 2645 x 602 is roughly 15. What is the longest wavelength of light capable.
To put it into your calculator correctly you need to use your ee button. A sample of copper with a mass of 635g contains 602 x1023 atoms calculate the mass of an average copper ato Get the answers you need now. Answer 1 of 3.
Scientific notation is also know as exponential notation. This website uses cookies to improve your experience analyze traffic and display ads. Math Expression Renderer Plots Unit Converter Equation.
Hit the EE button above the 7 also X -1 4. Up to 24 cash back 1. Scientific notation is also know as exponential notation.
Type in the exponent 23. 1 mole 602 X 10 23 things This is called Avogadros number PPTX from vdocumentsmx.

The Mole Concept What Is A Mole I In Chemistry A Mole Is A Counting Unit Abbreviated Mol 1 1 Mol 6 022x10 23 Representative Particles Avogadro S Ppt Download
What Is The Mass In Grams Of 6 022 X10 23 Atoms Of C Quora
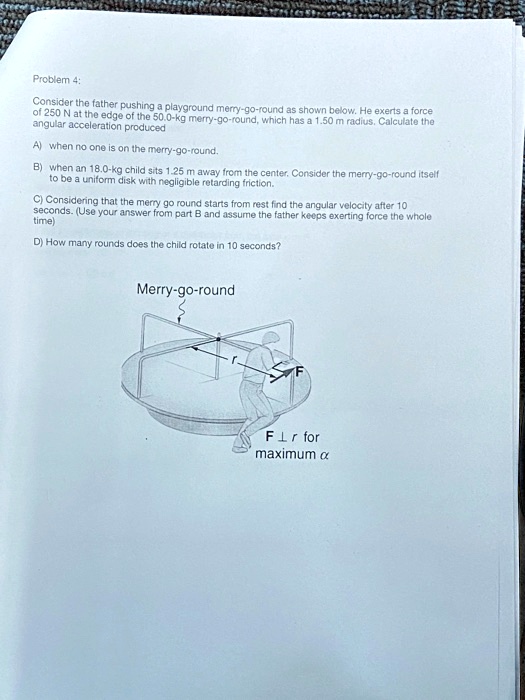
Browse Questions For Physics 101 Mechanics

The Mole Concept What Is A Mole I In Chemistry A Mole Is A Counting Unit Abbreviated Mol 1 1 Mol 6 022x10 23 Representative Particles Avogadro S Ppt Download

Mole It Is Just A Number 6 02x10 23 Part I Youtube

22 Proof Principle Of Mathematical Induction Mathgotserved 1 2 2 2 3 2 N 2 Nn 12n 1 6 Youtube

Browse Questions For Chemistry 101

The Mole Concept Avogadro S Number X Ppt Download
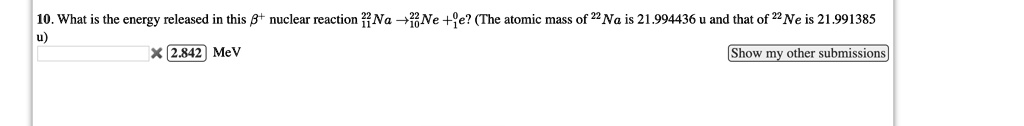
Browse Questions For Physics 101 Mechanics

How To Convert Atoms To Grams With A Calculator

Avogadro S Number Calculator

Mole Problems Solution Guide Ppt Download

The Mole Wongchemistry

Browse Questions For Physics 101 Mechanics
Calculator Help Griger Science

Doc 117 B P S Xi Chemistry Iit Jee Advanced Study Package 2014 15 By S Dharmaraj Issuu

1012 Chemistry Pdf Chemical Equilibrium Mole Unit